Aniline reacts with perchloric acid6/12/2023 
I kno to use the ph = pka + log (base/acid). What is the pH of a solution prepared by dissolving 8.40 g of aniline hydrochloride C6H5NH3Cl in 750 mL of 0.210 M aniline,C6H5NH2?.the density aniline c6h5nh2 is 1.02g/mL how many molecules are present in 1.00Lsample.I don't know where to start? Do I need an ICE table? Given that Ka for HIO is 3.2 × 10-11 at 25 ☌, what is the value of Kb for IO– at 25 ☌? Then, Given that Kb for C6H5NH2 is 1.7 × 10-9 at 25 ☌, what is the value of Ka for C6H5NH3 at 25 ☌?.Perchloric acid (HClO4) (aq) + tetraphosphorus decaoxide (s) -> phosphoric acid (aq) + 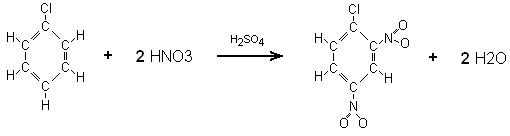
According to the following reaction, how many grams of tetraphosphorus decaoxide are required for the complete reaction of 26.5 grams of perchloric acid (HClO4)?.The equilibrium constant for the reaction of the weak base aniline is Kb = 4.3e-10.Ĭ6H5NH2 (aq) + H2O (l) -> C6H5NH3+ (aq) + OH- (aq).It takes on a H^+ from HClO4 and forms the salt. Written out, would the reaction between anihiline and perchloric acid look like this:Ĭ6H5NH2 + HClO4 -> C6H5ClO4 + HNH2 ? The NH2 group on aniline is basic.Written out, would the reaction between anihiline and perchloric acid look like this: C6H5NH2 + HClO4 -> C6H5ClO4 + HNH2 ?
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
